



INCRELEX® to Treat Severe Primary IGF-1 Deficiency
On this page

Olive, a former INCRELEX patient, at age 18
Mechanism of Action
INCRELEX® contains human insulin-like growth factor-1 produced by recombinant DNA technology (rhIGF-1). IGF-1 is a key hormonal mediator on growth stature. (1)
Established Safety Profile With Increlex
Adverse reactions occurring with Increlex in ≥5% of patients in clinical studies included* (1):
- Hypoglycemia, lipohypertrophy, bruising, otitis media, serous otitis media, snoring, tonsillar hypertrophy, headache, dizziness, convulsions, vomiting, hypoacusis, fluid in middle ear, ear pain, abnormal tympanometry, cardiac murmur, arthralgia, pain in extremity, thymus hypertrophy, ear tube insertion
Hypoglycemia was reported by 30 patients (42%) at least once during their course of therapy (1)
- Most cases of hypoglycemia were mild or moderate in severity
- Five subjects had severe hypoglycemia (requiring assistance and treatment) on one or more occasions and 4 subjects experienced hypoglycemic seizures/loss of consciousness on one or more occasions
- 14 of the 30 patients (47%) reporting hypoglycemia had a history of hypoglycemia prior to treatment
- The frequency of hypoglycemia was highest in the first month of treatment and episodes were more frequent in younger children
- Symptomatic hypoglycemia was generally avoided when a meal or snack was consumed either shortly (i.e., 20 minutes) before or after the administration of INCRELEX®
No patients withdrew from any clinical study because of adverse reactions. (1)
Improve Growth Rates in Children With Severe Primary IGF-1 Deficiency
In clinical trials, INCRELEX® improved statural growth in patients diagnosed with SPIGFD.(1)
Average height SDS had increased from start of treatment by +0.8 at Year 1 and +1.2 at Year 2.(1)
- The average height SDS was -6.7 at baseline for the 61 subjects in the efficacy analysis.
Mean height velocity nearly tripled over baseline in the first year (P<0.0001).(1)
- Mean height velocity increased to 8 cm/year in the first year, on average, from a baseline of 2.8 cm/year (P<0.0001).
- Mean height velocity was sustained at approximately 5 cm/year in Years 2 through 6 of treatment.
- Forty-nine subjects were included in an analysis of the effects of INCRELEX® on bone age advancement.
- The mean ± standard deviation (SD) change in chronological age was 4.9 ± 3.4 years.
- The mean ± SD change in bone age was 5.3 ± 3.4 years.
When Does Primary IGFD Become Severe Primary IGFD?
- height standard deviation score (SDS) ≤ -3.0
- basal IGF-1 SDS ≤ -3.0
- normal or elevated GH
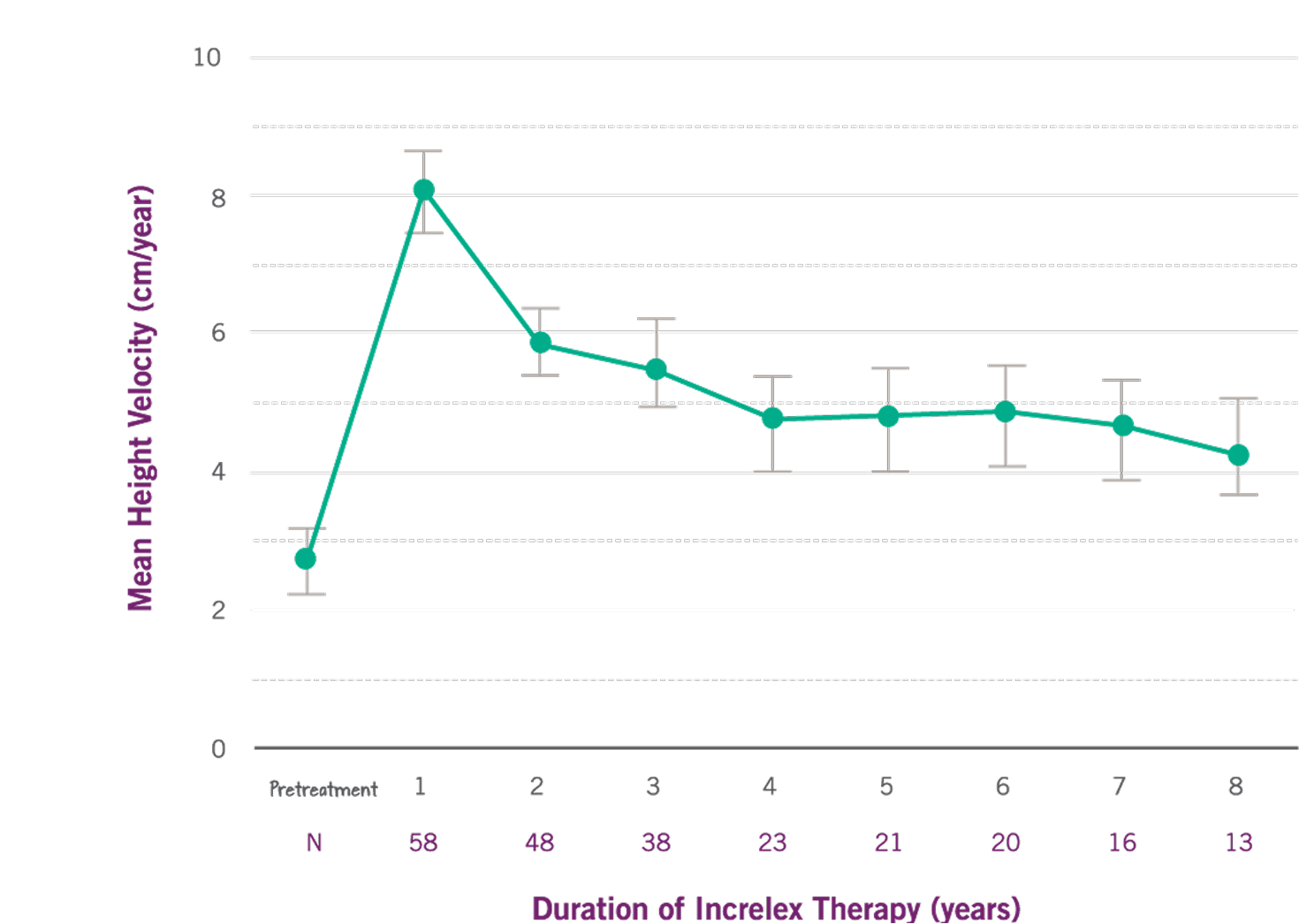
Growth Rate with INCRELEX® over 8 years (1)
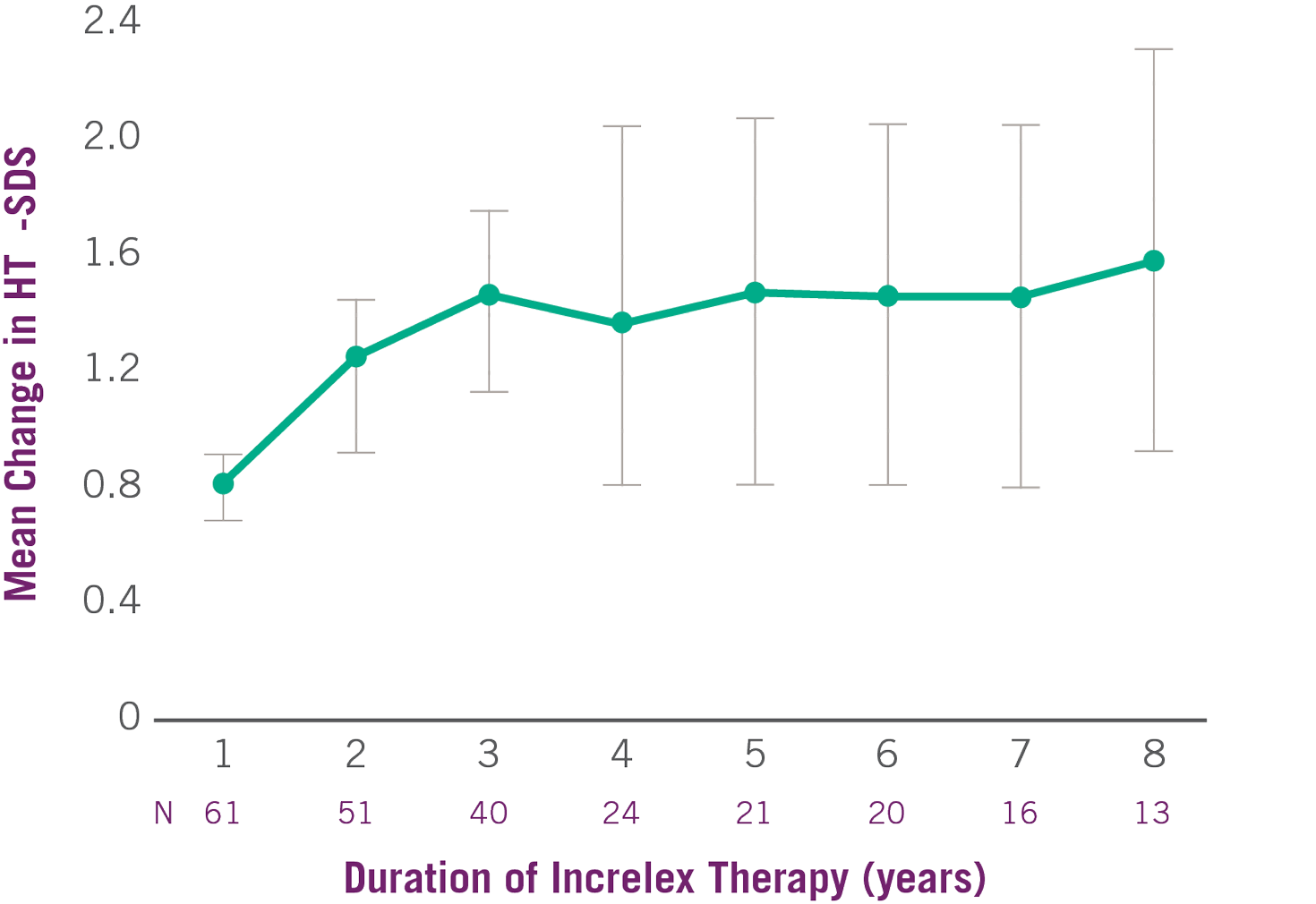
Change in Height SDS with INCRELEX® Over 8 Years (1)
INCRELEX® Dosing and Administration
The dosing of INCRELEX® should be individualized for each patient. This should be based on the periodic assessment of each patient’s weight, tolerability, and laboratory parameters. (1) Patients should start INCRELEX® treatment on a weight-based dose in the recommended starting dose range of 0.04 to 0.08 mg/kg twice daily, given subcutaneously. (1)

INCRELEX® Dosing is Weight-based (1)
Adapted from INCRELEX® full prescribing information. The following formula was used to calculate the dosage as units per injection: patient body weight (kg) x single dose of Increlex (mg/kg) x 1 mL/10 mg x 100 units/1 mL† = units/injection. Two doses per day, morning and evening before or after (plus/minus 20 minutes) a meal or snack (a separation of 12 hours) are recommended.1 †1 mL=1 cc.
Pre-prandial glucose monitoring is recommended at treatment initiation and until a well-tolerated dose is established. If frequent symptoms of hypoglycemia or severe hypoglycemia occur, pre-prandial glucose monitoring should continue. If hypoglycemia occurs with recommended doses despite adequate food intake, the dose should be reduced.1
Appropriate dose adjustments of INCRELEX® are necessary to avoid administering a dose that may be too low for an individual patient. Patients should be weighed and measured frequently, as regular monitoring is critical for proper unit dosing.1

Three Simple Steps to INCRELEX® Dosing (1)
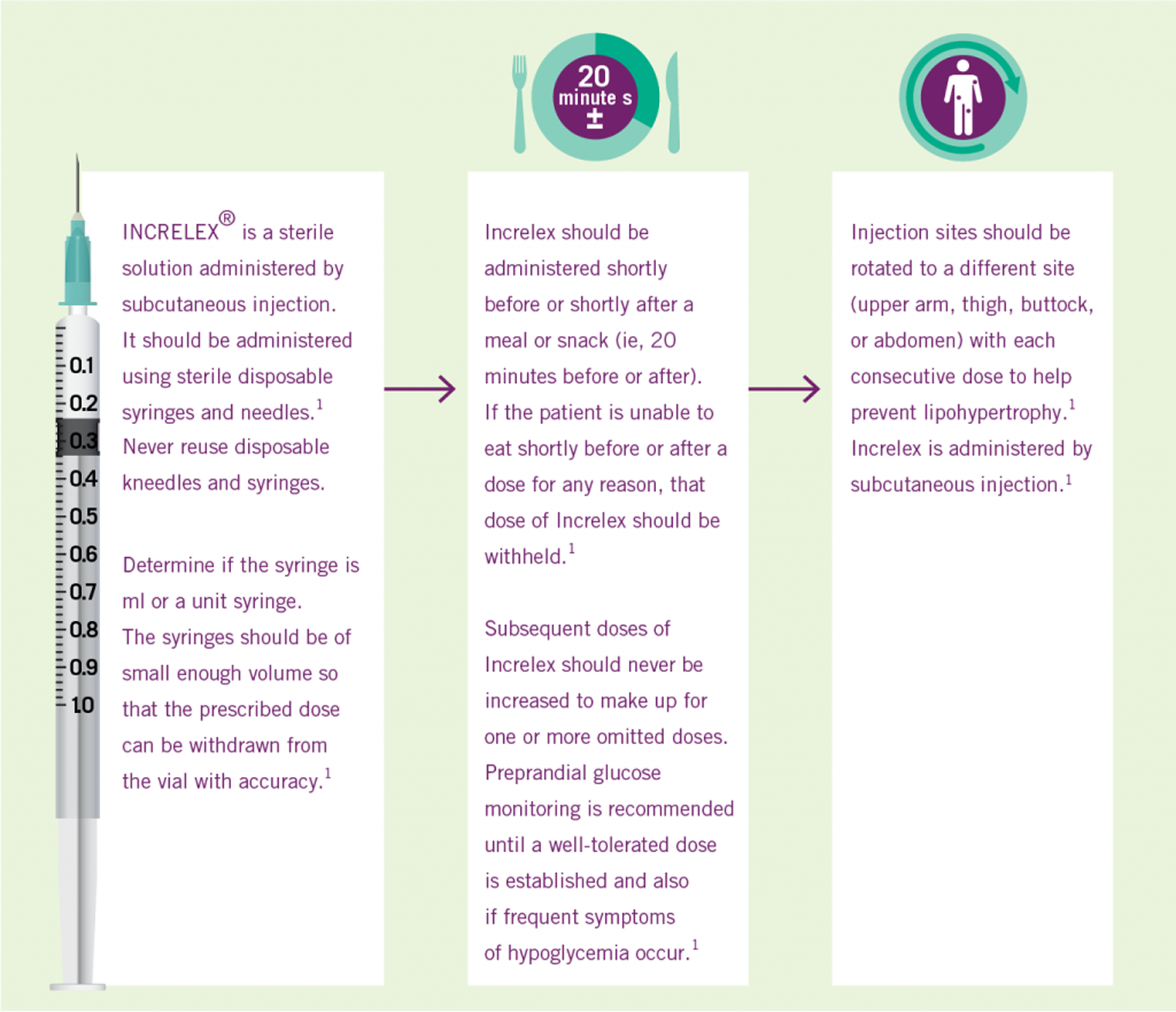
INCRELEX® Administration (1)
INCRELEX® Supply
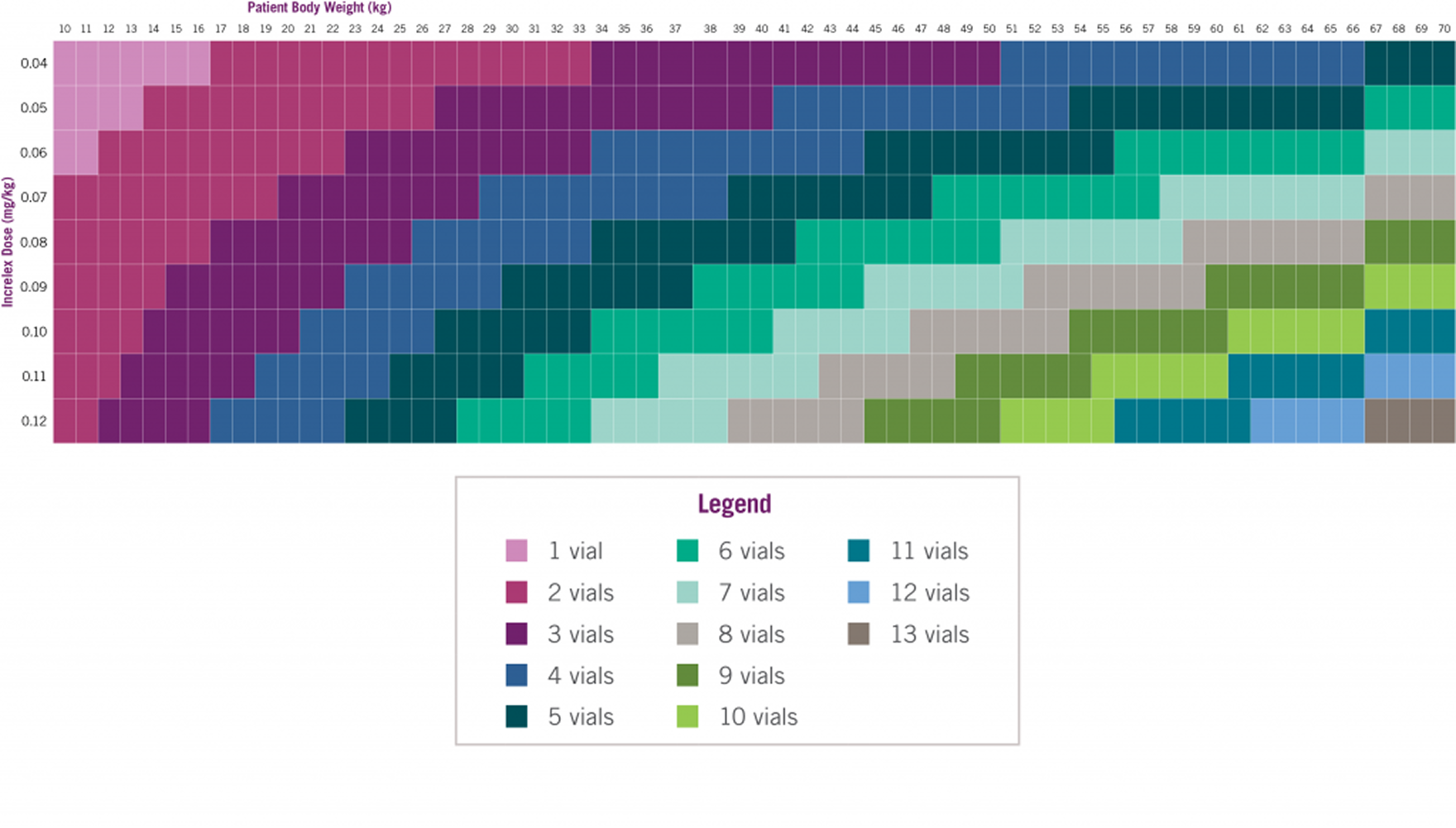
Number of INCRELEX® vials to prescribe for a 30-day supply*4
INDICATION
INCRELEX® (mecasermin) is a prescription medicine used in children 2 years and older with short stature that have severely low levels of the hormone, insulin-like growth factor-1 (IGF-1). IGF-1 is needed for normal growth.
INCRELEX is not a substitute for growth hormone. It cannot be used for children with short stature due to low levels of growth hormone, malnutrition, when their thyroid gland doesn’t make enough thyroid hormone, or when taking anti-inflammatory steroids used to manage inflammation.
IMPORTANT SAFETY INFORMATION
Always give INCRELEX exactly as your doctor directed.
Do not take INCRELEX if you are allergic to IGF-1 or any of its other ingredients. Report any allergic reactions.
INCRELEX should be injected under the skin (subcutaneous injection). It should not be injected directly into a blood vessel.
INCRELEX should not be used after growth plates close which happens during puberty.
INCRELEX should not be used in children with cancerous tumors or a history of cancer.
Hypoglycemia (low blood sugar): INCRELEX should be administered 20 minutes before or after a meal or snack and should not be administered when the meal or snack is skipped.
Checking blood glucose levels is recommended. The dose of INCRELEX may need to be adjusted until an appropriate dose is decided by your doctor.
Intracranial Hypertension: Increased pressure in your skull may occur because of cerebrospinal fluid buildup around your brain. Therefore, your doctor may require an eye examination at the start of INCRELEX treatment and periodically during the time you are taking INCRELEX.
Lymphoid Tissue Hypertrophy: Lymphoid tissue hypertrophy is a noncancerous increase in the number of immune cells called lymphocytes. Patients should have periodic examinations with your doctor to rule out potential complications.
Slipped Capital Femoral Epiphysis: Slipped capital femoral epiphysis is a bone problem where the top of the upper leg (femur) slips apart. This may lead to a serious condition where bone tissue dies due to a lack of blood supply (osteonecrosis). Get medical help for your child right away if your child develops a limp or has hip or knee pain.
Progression of Scoliosis: Your doctor will monitor you during treatment with INCRELEX if you have a history of scoliosis.
Malignant Neoplasia: There have been reports of cancerous tumors in children who received INCRELEX. It is unknown whether there is any relationship between INCRELEX therapy and new occurrence of tumors. Tumors were mostly reported in patients with rare genetic conditions of short stature associated with a higher risk of cancer, or in patients already at risk of cancer.
The tumors were seen more frequently in patients who received INCRELEX at higher than recommended doses or at doses that produced IGF-1 levels above normal for age and sex. Your doctor will carefully monitor you during your treatment with INCRELEX for development of tumors. If cancerous tumors develop, your doctor will stop your INCRELEX treatment.
Risk of Serious Adverse Reactions in Infants due to Benzyl Alcohol Preserved Solution: Serious and fatal adverse reactions can occur in neonates and infants treated with benzyl alcohol-preserved drugs. Use of INCRELEX in infants is not recommended.
The most common adverse reactions include low blood sugar, reactions at the injection site or throughout your body, and enlarged tonsils.
You are encouraged to report negative side effects of prescription drugs by contacting Eton Pharmaceuticals, Inc. at 1-855-224-0233 or the U.S. Food and Drug Administration (FDA) at www.fda.gov/safety/Medwatch or call 1-800-FDA-1088.
Please see full Prescribing Information for more information.
INDICATION
INCRELEX® (mecasermin) is a prescription medicine used in children 2 years and older with short stature that have severely low levels of the hormone, insulin-like growth factor-1 (IGF-1). IGF-1 is needed for normal growth.
INCRELEX is not a substitute for growth hormone. It cannot be used for children with short stature due to low levels of growth hormone, malnutrition, when their thyroid gland doesn’t make enough thyroid hormone, or when taking anti-inflammatory steroids used to manage inflammation.
IMPORTANT SAFETY INFORMATION
Always give INCRELEX exactly as your doctor directed.
Do not take INCRELEX if you are allergic to IGF-1 or any of its other ingredients. Report any allergic reactions.
INCRELEX should be injected under the skin (subcutaneous injection). It should not be injected directly into a blood vessel.
INCRELEX should not be used after growth plates close which happens during puberty.
INCRELEX should not be used in children with cancerous tumors or a history of cancer.
Hypoglycemia (low blood sugar): INCRELEX should be administered 20 minutes before or after a meal or snack and should not be administered when the meal or snack is skipped.
Checking blood glucose levels is recommended. The dose of INCRELEX may need to be adjusted until an appropriate dose is decided by your doctor.
Intracranial Hypertension: Increased pressure in your skull may occur because of cerebrospinal fluid buildup around your brain. Therefore, your doctor may require an eye examination at the start of INCRELEX treatment and periodically during the time you are taking INCRELEX.
Lymphoid Tissue Hypertrophy: Lymphoid tissue hypertrophy is a noncancerous increase in the number of immune cells called lymphocytes. Patients should have periodic examinations with your doctor to rule out potential complications.
Slipped Capital Femoral Epiphysis: Slipped capital femoral epiphysis is a bone problem where the top of the upper leg (femur) slips apart. This may lead to a serious condition where bone tissue dies due to a lack of blood supply (osteonecrosis). Get medical help for your child right away if your child develops a limp or has hip or knee pain.
Progression of Scoliosis: Your doctor will monitor you during treatment with INCRELEX if you have a history of scoliosis.
Malignant Neoplasia: There have been reports of cancerous tumors in children who received INCRELEX. It is unknown whether there is any relationship between INCRELEX therapy and new occurrence of tumors. Tumors were mostly reported in patients with rare genetic conditions of short stature associated with a higher risk of cancer, or in patients already at risk of cancer.
The tumors were seen more frequently in patients who received INCRELEX at higher than recommended doses or at doses that produced IGF-1 levels above normal for age and sex. Your doctor will carefully monitor you during your treatment with INCRELEX for development of tumors. If cancerous tumors develop, your doctor will stop your INCRELEX treatment.
Risk of Serious Adverse Reactions in Infants due to Benzyl Alcohol Preserved Solution: Serious and fatal adverse reactions can occur in neonates and infants treated with benzyl alcohol-preserved drugs. Use of INCRELEX in infants is not recommended.
The most common adverse reactions include low blood sugar, reactions at the injection site or throughout your body, and enlarged tonsils.
You are encouraged to report negative side effects of prescription drugs by contacting Eton Pharmaceuticals, Inc. at 1-855-224-0233 or the U.S. Food and Drug Administration (FDA) at www.fda.gov/safety/Medwatch or call 1-800-FDA-1088.
Please see full Prescribing Information for more information.
